 It is a fiddle to get a formula for the periodicity of the emission lines of hydrogen. Periodicity of hydrogen spectral lines.The development of these harmonics was as follows. Much less evidence-based are the shell regions, which are defined by the use of special Spherical harmonic. It states that electrons in an atom differ in at least one quantum number. So we have the Aufbau rule and Madelung's rule, and so on to estimate the filling of orbitals.Ībsolutely fundamental is the Pauli exclusion principle. This results from empiricism in chemistry. During bonding with another atom, the shape of the volumes changes and the electrons can shift their position a little. 
Using this equation to describe the electron arrangement of other elements does not reflect the reality of chemical compounds.īound electrons occupy a certain volume in the atom and can be considered immobile. Schrödinger's equation can only describe the excited states in the hydrogen atom and in hydrogen-like ions. I have also wondered that Schrodinger's equation is solvable for 1-electron atoms, yet chemists have extended the electron configurations to large and heavy elements What is the experimental evidence of orbitals and orbital filling orders in heavier elements beyond hydrogen? 
The shell structure of atoms is indeed mentioned via X-ray studies. I have never seen any chemistry or physical text that shows or discusses experimental evidence of electron configurations. Note that Bohr came up with electron configurations based on the chemical properties of elements. 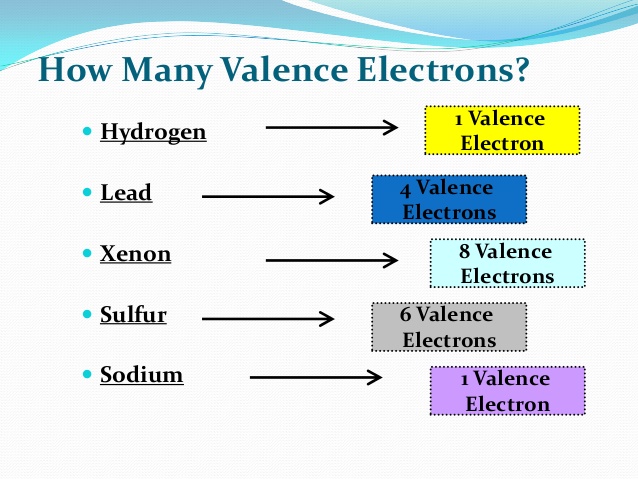
The question from the point of view of physics is if you happen to see the electron configuration of Pb as 4f $^$, what is the experimental evidence that lead's electron configuration is like that and this is the correct orbital order. Nevertheless, the orbital concept has been extended to larger atoms even with hundreds of electrons. Mullikan had coined the term orbitals as a short form of one-electron wavefunctions, but obviously, all the periodic table elements are not one electron atom after hydrogen. So we have the Aufbau rule and Madelung's rule, and so on to estimate the filling of orbitals. I have also wondered that Schrodinger's equation is solvable for 1-electron atoms, yet chemists have extended the electron configurations to large and heavy elements like lead or mercury. The author has mainly objected that orbitals are not physical objects, but mathematical constructs, and perhaps chemists take them too literally. In 1990, the Journal of Chemical Education (American Chemical Society) published a little bit controversial article titled " The nature of the chemical bond-1990: There are no such things as orbitals! J. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
